Advanced Oxidative Processes
The search for advanced oxidation technologies intensified in the 1990s, along with the emerging concern about the presence of recalcitrant pollutants in water and air (pharmaceuticals, hormones, pesticides, etc.). In this context, an Advanced Oxidation Process (AOP) is defined as any oxidative process in which the hydroxyl radical is the dominant species.
For an oxidation reaction to occur, it must have high free energy (or electrical potential), i.e. it must be thermodynamically favorable. Table 1 shows the reduction potentials of some of the most appropriate oxidizers for environmental applications.
Table 1 – reduction potential of common oxidizing agents.
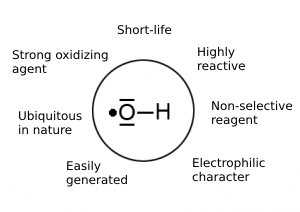
The hydroxyl radical has a reduction potential of 2.80 V, and is thus a strong oxidizing agent. It is a non-selective, short-lived radical that reacts with organic compounds by several mechanisms. This radical is naturally present in reactions that occur, for example, on river surfaces, in the atmosphere, and in the human body (aging process).
Figure 1 – characteristics of the hydroxyl radical [1].
The mechanisms involved in hydroxyl radical-mediated reactions are explained by transition state theory, which provides theoretical guidance for finding the most efficient AOP. Transition state theory reveals that hydroxyl radicals can accelerate the rate of oxidation of organic compounds to orders of magnitude higher than that of regular oxidants. This occurs due to the fact that radical reactions have considerably lower activation energies than non-radical reactions. Thus, through the generation of these radicals, oxidizing agents such as O2, H2O2, and O3 have their reactions catalyzed when combined with transition metals, oxides, photons, ultrasound, and other technologies. From these combinations arise the advanced oxidation technologies, as illustrated in table 2.
Table 2 – advanced oxidation technologies and processes [1].
In this context, the understanding of the mechanisms involved in oxidation reactions and the search for optimization of advanced oxidation technologies constitute a scientific frontier of enormous importance.
References:
1- OPPENLÄNDER, T. Photochemical Purification of Water and Air. Advanced Oxidation Processes Principles, Reaction Mechanisms, Reactor Concepts. GbH &Co. KGaA, Weinheim: 2003 WILEY-VCG Verlag. 368.
2- TANG, W. Z. Physicochemical Treatment of Hazardous Wastes. CRC Press, Boca Raton, Florida, 2000.



 Imprimir
Imprimir